ANCA-associated vasculitis
Information for patients from the Renal Department
This leaflet is for people coming to East Kent Hospitals, with suspected or confirmed ANCA-associated vasculitis.
Vasculitis means inflammation of the blood vessels. It can affect any of the body’s blood vessels, causing a variety of different symptoms and problems.
ANCA is short for ‘anti-neutrophil cytoplasmic antibody’. ANCA is linked with two particular types of vasculitis that commonly affect a person’s kidneys.
What is ANCA-associated vasculitis?
ANCA are often found, and have a direct role in causing blood vessel inflammation, in two types of vasculitis:
granulomatosis with polyangiitis (GPA or c-ANCA vasculitis; previously known as Wegener’s granulomatosis); and
microscopic polyangiitis (MPA or p-ANCA vasculitis).
What causes ANCA-associated vasculitis?
The exact cause of ANCA-associated vasculitis is not known. However, in some people an environmental trigger or infection may cause symptoms.
What are the symptoms?
Around 7 in 10 people with GPA, and 9 in 10 of those with MPA will have inflammation affecting their kidneys. The effect on patients can range from having:
small amounts of invisible blood and protein in your urine (with normal kidney function)
through to complete kidney failure.
As well as affecting the kidneys, GPA and MPA may also affect other parts of the body.
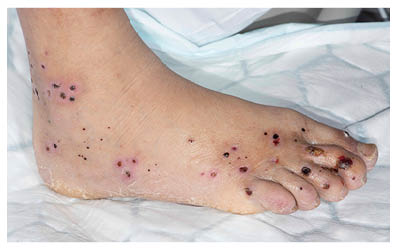
Skin, typically with a ‘purpuric’ rash (see image). This is a purplish discolouration of the skin.
Lung symptoms can include coughing and breathlessness. Sometimes people will cough up blood with these conditions.
Upper respiratory tract (ears, nose, throat) symptoms include:
nose bleeds
runny nose
nasal and sinus congestion; or
pain and reduced hearing.
These symptoms are more common in people with GPA.
Nervous system. Usually the peripheral nerves are affected. These are the nerves that exit your spinal cord and connect to your muscles and skin. If affected this commonly causes numbness in your hands and feet, and occasionally muscle weakness.
Eyes, for example inflammation of the white of the eye.
Joints and muscles, typically aching or pain.
General symptoms. It is common for people with GPA and MPA to have some or all of the following symptoms, which can continue despite treatment.
weight loss
fevers and sweats; and
fatigue (extreme tiredness).
In general, the treatment for the vasculitis affecting your kidneys will also work in treating the effects listed above. However, sometimes we will need to involve other specialists in your care. These can include respiratory specialists, neurologists, rheumatologists, and others.
How is ANCA-associated vasculitis diagnosed?
It is quite common for the diagnosis not to be suspected in people whose early symptoms have a more common cause - for example, cough, sinus problems, or hearing changes.
Doctors will usually make a provisional diagnosis of GPA or MPA in people who have the symptoms listed above, including evidence of kidney damage (blood and / or protein in the urine, with or without blood tests showing impaired kidney function) and an abnormal ANCA blood test. This blood test can be arranged by both GPs and hospital specialists.
Other investigations are usually carried out to confirm a positive diagnosis of GPA or MPA. In people who are thought to have vasculitis affecting their kidneys - usually due to blood tests showing impaired kidney function or urine test showing blood or protein - a kidney biopsy would usually be needed. For more information, see our information sheet Having a kidney biopsy: your questions answered.
Other investigations may be carried out if other organs are affected. This may include a lung CT scan, nerve conduction studies, and a skin biopsy. Sometimes these investigations will change the original diagnosis of GPA or MPA, and may show a different diagnosis.
How is ANCA-associated vasculitis treated?
Although GPA and MPA are regarded as two different diseases, our approach to treatment is mainly the same for each condition. The main treatment involves using medicines that suppress the body’s immune system and stop production (or remove) ANCA and other molecules that cause inflammation, organ damage, and symptoms.
We would expect most people with symptoms attributable to their ANCA vasculitis to begin to feel better within a few weeks of starting treatment. In those with impaired kidney function, we expect to see kidney function stabilise within two to four weeks. By three to six months most people’s kidney function will have improved as much as it is going to. There are three different stages in the treatment of GPA and MPA, and all patients will progress through at least the first two phases.
The induction phase involves treatment with very strong immune suppressing medications, typically with intravenous (IV) cyclophosphamide infusions or Rituximab (see below). This phase usually lasts three months, but it can last up to six months if needed. The aim of treatment is to quickly suppress inflammation to prevent organ damage and to allow the healing of damaged tissues. During this period people are at greatest risk from infection. We aim to minimize this risk by:
keeping the induction phase as short as possible
using the lowest dose of steroids, or replace steroids with avacopan, since high doses of steroids are one of the main factors increasing the risk of infection
using additional medications: antibiotics and antifungals
closely monitoring your response to treatment and any adverse effects that may arise, including infection. Before each intravenous (IV) cyclophosphamide infusion, blood tests are taken, and you will be seen in our clinic or on our day case unit by one of our consultants, usually with the second, fourth, and last dose of IV cyclophosphamide.
Maintenance phase: more ‘gentle’ immune suppression is introduced following the induction phase, once our doctors are sure that the induction treatment has fully suppressed inflammation and stopped on-going damage. This treatment, being less ‘toxic’, has fewer side effects and is more suitable for long term use. The aim is to prevent inflammation starting up again - known as a relapse. Depending on your type of ANCA-associated vasculitis (GPA or MPA) and other factors that will be discussed with your consultant in the clinic, we may recommend withdrawal of immunosuppression after two to three years. In some people, especially those whose vasculitis relapses, we may recommend continuing immunosuppression over the long term.
Immune suppression withdrawal phase. Some people – usually those who have not suffered a relapse during the maintenance phase – may have their immune suppressing medication carefully reduced and stopped after a period of time, usually a minimum of two to three years from the start of their induction treatment. Overall half of people with GPA and MPA will eventually have a relapse (although this is usually mild) and so doctors often keep patients with GPA and MPA under very long term but infrequent review.
In addition to the above phases, people who have severe kidney involvement may need other treatments, including dialysis (for those with complete kidney failure) and plasma exchange which removes preformed ANCA from the blood.
If you have any questions about your treatment, our doctors, nursing staff, and pharmacists will be more than happy to answer them.
Why have I been given a patient diary?
When you start treatment for vasculitis you will be given a patient diary that will provide you with information on:
when you are due to attend our day case unit, for treatments such as intravenous cyclophosphamide infusions
when you are due to come to the clinic to be reviewed by one of our doctors
when you need to have blood tests done; and
what dose of steroid (prednisolone - see below) you need to take each week.
What medicines are used to treat ANCA-associated vasculitis, and what side effects do they have?
Two of the medicines we use to treat ANCA-associated vasculitis (Cyclophosphamide and Rituximab) are given as IV infusions, usually on one of our day case treatment units at Kent and Canterbury Hospital (for kidney (renal) and neurology patients) or Ashford and Margate for other patients. The remaining medicines are tablets which patients take at home and to start with, we will usually provide a 14 to 28 day prescription. Usually we will ask your GP if they are able to take over prescribing after this time, but sometimes we will continue to issue prescriptions if your GP is unable to.
The treatments and medicines that we recommend for ANCA-associated vasculitis are based on research studies (usually gold-standard ‘randomized controlled trials’) conducted over the last 25 years or so. East Kent Hospitals have been actively involved in some of these studies over the last 10 years. The findings of these studies allow us to advise our patients on the best medicine at each stage of their treatment, and provide patients with information on benefits, risks, and alternatives. Some areas of uncertainty remain, for example how long to continue maintenance treatment for. We will explain where such uncertainty exists and, on occasions, we may invite people to participate in research studies that aim to answer remaining questions about the treatment of ANCA-associated vasculitis.
If you have questions about your condition or its treatment or you experience side effects, it is usually best to discuss these with us when you attend clinic or our day case units for infusions. During normal office hours(usually Monday to Friday 8am to 4pm) you can also phone your consultant via our secretaries. If we are unavailable a message can be left and we will call you back.
If you are experiencing severe side effects out of office hours you should contact your GP out-of-hours service or NHS 111 for advice. In an emergency, go to your nearest Emergency Department.
Steroids (prednisolone tablets and intravenous methylprednisolone)
Steroids (prednisolone tablets and intravenous methylprednisolone)
Steroids are a highly effective treatment for inflammation and have been used in the treatment of vasculitis for years.
They have a wide variety of side effects, most of which relate to the dose and how long the steroids are taken for. For this reason, we use higher doses of steroids to start with, and aim to quickly reduce the dose over eight to 12 weeks, and then use a lower dose – usually 2.5 to 5mg – for the next 12 months. Although usually taken as a tablet, occasionally intravenous (injected) steroids (methylprednisolone) are used in people with severe disease.
Common side effects of steroids are:
General: difficulty sleeping; effects on mood (happiness and low mood); tremor / shakiness / agitation; weight gain; change in appearance (‘moon face’).
Skin: easy bruising; stretch marks; wounds may be slower to heal.
Patients are more likely to get an infection, including fungal infections* (such as candidiasis – thrush).
Development of diabetes: this is often in patients who are already more likely to develop diabetes due to being overweight, or having a family history. This may be reversible when steroids are reduced or stopped.
Musculoskeletal system: long term steroid usage may lead to loss of bone strength, which in turn increases risk of bone fractures*; loss of muscle strength (proximal myopathy).
Eyes: glaucoma (high pressure within the eyes); acceleration of cataract formation. If you are under follow-up with an optician / optometrist you may wish to tell them that you are taking steroids.
Gastrointestinal: indigestion; ulceration of the stomach or duodenum*; pancreatitis.
*We prescribe additional medications to counteract these side effects.
Avacopan (Tavneos®)
Avacopan is a new treatment for ANCA-associated vasculitis. It was approved for use in the UK in late 2022, following publication of the ADVOCATE study. Avacopan tablets may be used as an alternative to prednisolone in selected patients with severe ANCA-associated vasculitis. Treatment usually lasts 12 months.
Common side effects of avacopan are (with approximate frequencies in the ADVOCATE study):
Nausea (feeling sick) (23%), diarrhoea, and vomiting (both 15%).
Sore throat / nasal cavities (15%).
Reduction in white blood count (19%), upper respiratory tract infection (15%), pneumonia (lung infection, 5%)
Headaches (20%) and abnormalities in liver function tests (5%). These will often be mild, but may occasionally need you to stop taking avacopan.
When starting avacopan we will treat people with a short course of steroids as well. This will usually be for the first two to three weeks. We monitor people receiving avacopan with regular blood tests, including full blood counts and liver function tests. These are usually done two, four, and eight weeks after starting avacopan, and every two to three months after that. This makes sure that low white blood counts or liver function test abnormalities are found early. Avacopan will be prescribed by the hospital, and is sent out by a specialist home delivery company.
Cyclophosphamide
Like steroids, Cyclophosphamide (or CYC for short) has been used to treat vasculitis for many years and is highly effective at switching off ANCA production and bringing inflammation under control quickly. Cyclophosphamide was originally developed to treat cancer and, whilst many of the side effects of CYC are those typically associated with chemotherapy, these are usually mild and CYC is often well tolerated in people with vasculitis. Generally, CYC is given as an intravenous infusion on six occasions over a three month period in one of our day case units.
Common side effects of CYC are:
Bone marrow suppression, resulting in anaemia and/or low white blood counts.
Predisposition to infection, including with the atypical organism Pneumocystis (or PCJ)*; the risk of infection is present even in those without white blood cell suppression.
Hair thinning or loss.
Inflammation in the bladder, causing blood in the urine*.
Nausea (feeling sick) and / or vomiting is fairly uncommon*.
*We prescribe additional medications (such as Mesna) to help with these side effects. We also monitor people receiving CYC with regular blood tests to make sure side effects are found early.
CYC inhibits sperm formation (spermatogenesis) and for women, may result in their monthly periods stopping, which can be permanent and result in premature ovarian failure. If you are planning to have more children, we will discuss alternative treatments to CYC with you, for example Rituximab.
CYC is also associated, over the long term, with an increased risk of bladder cancer and non-melanoma skin cancers. These side effects are generally associated with the total cumulative dose of CYC a person receives over their lifetime, with a doses below 25g being considered to be acceptable. A standard three month course of CYC will not exceed 7.2g, so we are confident that our approach does not increase a person’s risk of bladder cancer. In people with a previous history of bladder cancer, we will discuss alternative treatments to CYC with you, for example Rituximab.
Azathioprine
Azathioprine (AZA for short) works in a similar way to CYC but is not associated with some of the possible side effects seen with CYC. It generally replaces CYC during the maintenance phase of treatment. AZA is taken as a tablet.
Common side effects of AZA are:
Bone marrow suppression, resulting in anaemia and/or low white blood counts.
Predisposition to infection - the risk of infection is present even in those without white blood cell suppression.
Nausea (feeling sick), loss of appetite, and vomiting.
Inflammation of the pancreas (pancreatitis); the precise risk is not known but is probably less than one in 100 patients; affects how the liver works.
Squamous cell cancer of the skin. The risk is associated with longer term use of AZA (often over 10years) and can be helped by limiting the amount of time a patient spends in direct sunlight by using hats and sunscreen.
We monitor people receiving AZA with regular blood tests (for example full blood counts and liver function tests) to make sure that side effects are found early. AZA will usually be prescribed by the hospital when you come to your appointments. Some GP surgeries may agree to take over prescribing AZA, but this is their decision.
Mycophenolate Mofetil (MMF)
Originally developed to prevent rejection of transplanted organs, Mycophenolate Mofetil may be used as induction treatment for milder MPA or as maintenance treatment for people who get side effects with Azathioprine. MMF is taken as a tablet/capsule.
Common side effects of MMF are:
Bone marrow suppression, resulting in anaemia and/or low white blood counts.
Predisposition to infection - the risk of infection is present even in those without white blood cell suppression.
Diarrhoea, abdominal pain, nausea (feeling sick), and vomiting.
We monitor people receiving MMF with regular blood tests (for example full blood counts and liver function tests) to make sure that side effects are found early. MMF will usually be prescribed by the hospital when you come to your appointments. Some GP surgeries may agree to take over prescribing MMF, but this is their decision.
Rituximab
Rituximab is a monoclonal antibody that targets cells in your bone marrow that normally produce antibodies. Because it is a large, complex protein molecule it is given by intravenous infusion; usually two doses separated by two weeks, then every six months for up to two years. It can be associated with infusion reactions. Rituximab is often used in people who have had a severe relapse of their vasculitis, or who cannot receive Cyclophosphamide.
Common side effects of Rituximab are:
Predisposition to infection.
Infusion reactions* may display as fevers, chills, shaking, headache, high temperature, and very rarely, breathing difficulties. Our experience, based on a systematic audit of people receiving Rituximab, suggests infusion reactions are much less common than the product literature suggests.
Nausea (feeling sick).
Itching, rash.
Low white blood cell counts.
Low levels of immunoglobulin G (IgG), where IgG levels drop following Rituximab infusion. This effect is often temporary; even if prolonged, very low levels of IgG that are likely to be associated with an increased risk of infection are uncommon and, where this does happen, can be treated with immunoglobulin infusions.
*We prescribe additional medications to counteract these side effects. We also monitor people receiving Rituximab with regular blood tests to make sure side effects are found at an early stage.
What about vaccinations?
Some treatments for vasculitis may affect response to certain vaccinations in the short term (up to six months). This is especially true for Rituximab, so before starting Rituximab treatment we will write to your GP and ask them to give you the following vaccinations.
Tetanus and polio boosters, if needed.
Pneumococcus, unless you have received this in the last five years; but there is no contraindication to vaccinate again within this period if your vaccination status is unknown.
Influenza, if during or before ‘flu season.
Hepatitis B would usually be a booster vaccination and only in at risk groups who would normally receive this, for example healthcare workers.
It is recommended that Rituximab treatment should be delayed for two to four weeks following your vaccinations, to allow the vaccinations to work effectively. Where Rituximab treatment needs to start urgently, we may arrange to give you these vaccines at our day case unit. Alternatively we may advise that the risks of delaying treatment until you have the vaccination outweighs any benefit the vaccinations may provide.
The following live-attenuated vaccinations should not be given to people who have had Rituximab, until at least six months after their treatment course has finished.
BCG
Yellow fever
Mumps-Measles-Rubella (MMR)
Oral polio
Varicella / herpes zoster (Shingles vaccine).
Inactivated or component vaccines can be given safely, but may be of reduced success. This includes vaccinations against Covid-19 (SARS-CoV-2). The British Society of Rheumatology recommends that, where possible, Rituximab treatments are scheduled at least four weeks after a Covid-19 vaccination.
Who should I contact if I have a question or concern about my condition or medication?
If you have any questions about your treatment, please speak to one of our doctors, nurses, or pharmacists; they will be more than happy to answer your questions.
Further information
For a more general view of vasculitis further information can be found on the Vasculitis UK or Versus Arthritis websites. Vasculitis UK also maintain an active peer-to-peer private support group on Facebook which some people may find very helpful.